Good News: Singapore Immediate Placement for Class B & C with 1 Reference Regulatory Approval
- ARQon

- Dec 17, 2018
- 2 min read
In recent ASEAN meeting, Singapore HSA has reiterated the country's initiative to enhance the facilitation of immediate market access. With just 1 reference regulatory approval, companies can place the product in the market with immediate approval (Lead Time: 0 days) after submission. Manufacturers are still subjected to questions raised by the authorities on compliance documentations while the product is still in the market.
The table below shows the most recent regulation regarding the IBR:

New immediate class C registration route for Class C Standalone Mobile Applications has been released by the HSA, see the table below for the conditions required:
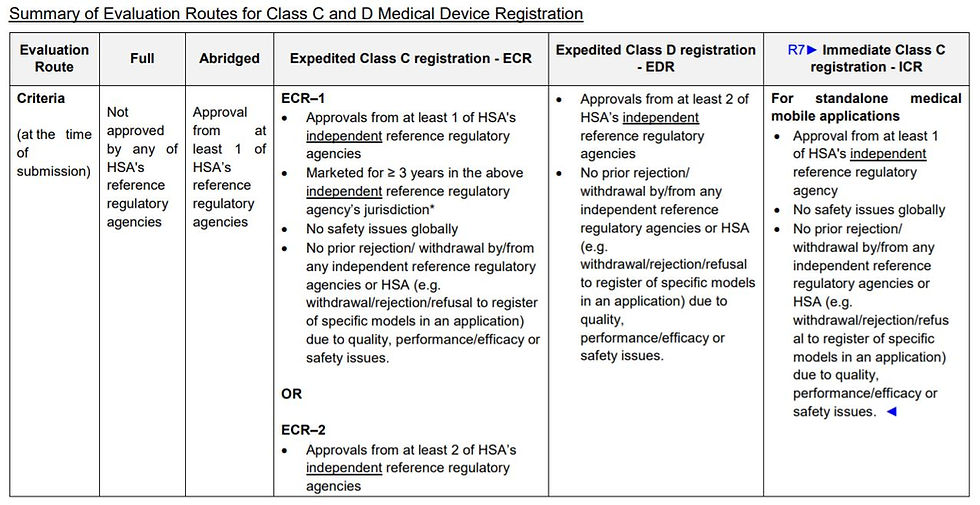

Both class B and C standalone medical mobile applications can be subjected to the immediate registration route so long as there is one reference regulatory agency approval and no safety issues globally.
With the increase in Telehealth products, HSA has also released clarifications on the scope of Telehealth devices that will be subjected to regulatory control. Telehealth medical devices are those that are intended to be used for investigation, detection, diagnosis, monitoring, treatment or management of any medical condition, disease, anatomy or physiological process, whereby the intended use is determined by the manufacturer.
Otherwise, if the manufacturer intends the devices or software to be used:
Solely to enable/encourage the user to adopt or maintain a healthy lifestyle; or for the user's general well-being; but
not to be used for any medical purpose
e.g. Fitbit watches, heart rate measuring devices for fitness purposes.
These devices will be considered as Wellness device, which can include the category of Telehealth products not intended for medical purposes. Wellness devices are not subjected to medical device regulatory controls if it is labelled as not for medical purpose and a clarification statement is provided on the device's presentation and advertisements on its non-relation to a medical device.
For more information, check out the following link:
Contact us at info@arqon.com.
.png)



Comments